Timeline of atomic theory4/3/2024 
"Rutherford Model of the Atom.", Gold Foil Experiment. "HISTORY OF THE ATOM FROM DEMOCRITUS TO BOHR AND SCHRÖDINGER." HISTORY OF THE ATOM FROM DEMOCRITUS TO BOHR AND SCHRÖDINGER. Dalton thought that atoms were the smallest units of matter tiny, hard spheres that could not be broken down any further. However, one of his underlying assumptions was later shown to be incorrect. "Joseph John Thomson." Homepage of the Chemical Heritage Foundation. Daltons ideas proved foundational to modern atomic theory. "John Dalton." Homepage of the Chemical Heritage Foundation. "From Quanta to Quarks." Senior Physics and Religion. Also, you can share the designs on different social media platforms, like Facebook, Twitter, LinkedIn, or Line."About This Artwork." Democritus, the Laughing Philosopher. You can further export the timeline in multiple formats, like Graphics, JPEG, PDF, or HTML. Once your required timeline is completed, you can share it amongst your colleagues or clients using the easy export and share option. Based on your research, you can also add or remove the sub-branches from the mind map. Alternatively, you can simply click on ""+"" in EdrawMax Online canvas to create a diagram of your preference.Ĭustomize your timeline by changing the color or adding more relevant data. In this case, you will find the Timeline Diagram under the ""Basic Diagram"" section under the ""General"" diagram types. I gave them this set of instructions to create an atomic theory timeline foldable. Select a pre-designed template by entering the Keyword in the ""Search"" section or exploring different diagram sets. We didn’t have time to cover every theory in class, so this project helped round-out our understanding of atomic theory. If this is your first time accessing the tool, you can create your personalized account from your personal or professional email address.ĮdrawMax Online comes with hundreds of free diagram templates. Log in EdrawMax Online using your registered email address. Learn how the atomic model has changed over time from ancient Greek thinkers to modern scientists. csv file, or you can also import media content, like images, icons, or symbols, right from your Google Drive or DropBox.  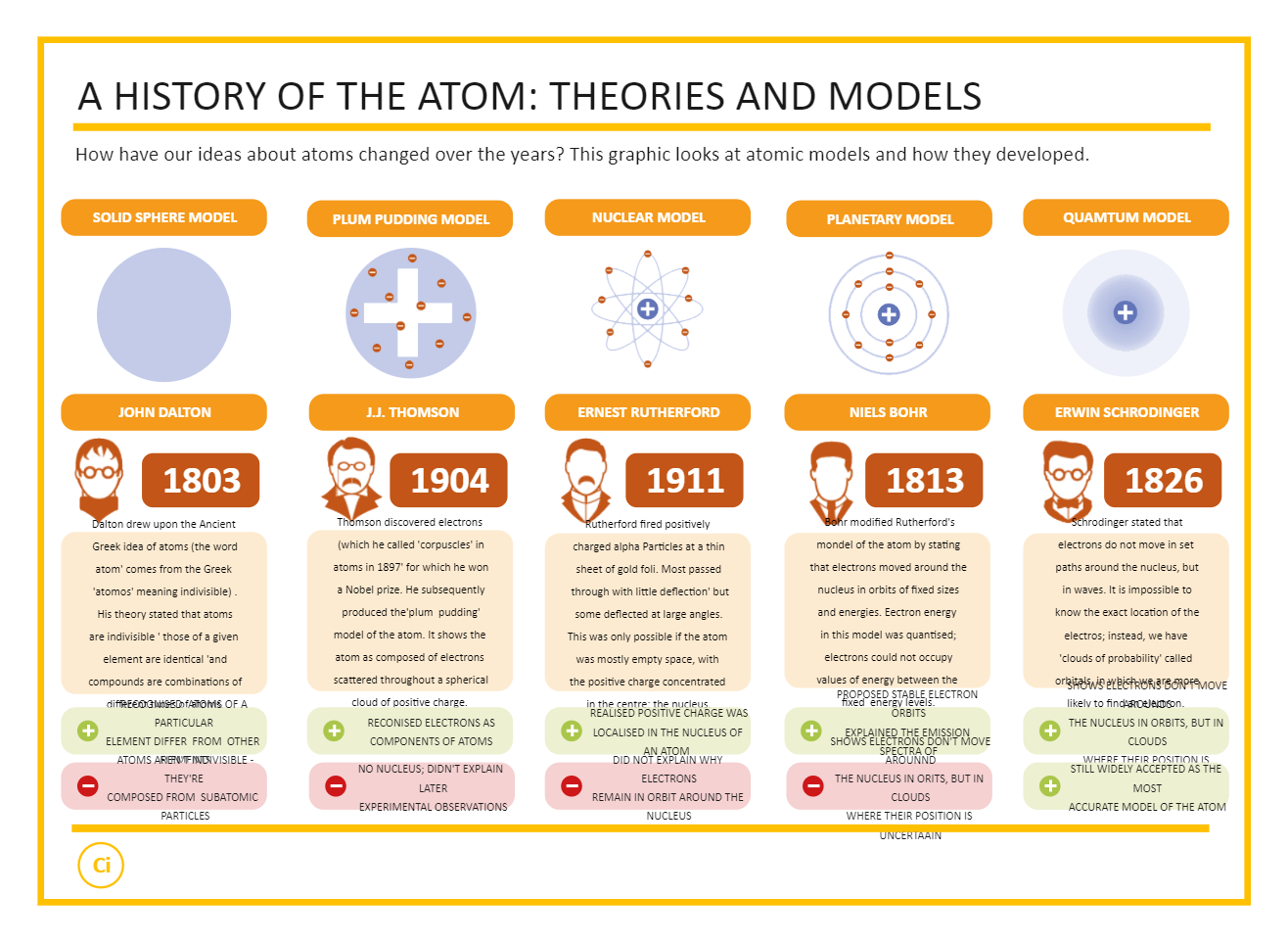
The timeline maker lets you import the data right from the. The free timeline maker has several features as you can instantly import the images or icons from your system or Google Drive or DropBox. In 1926, Erwin Schrödinger denied Bohr's theory and stated, you can not confirm the location of electrons inside the atoms.Ĭreating a timeline in EdrawMax Online is pretty simple. Neil Bohr's theory about atoms in 1913 showed that the electrons stay in orbitals of fixed size and energy level. Attempts to trace precisely how Dalton developed this theory have proved futile even Dalton’s own recollections on the subject are incomplete. The law of multiple proportions states that if two elements form more than one compound between them, the masses of one element combined with a fixed mass of the second element form in ratios of small integers. John Dalton - Atomic Theory, Chemistry, Physics: By far Dalton’s most influential work in chemistry was his atomic theory. The modern-day application of chemists quest to refine and purify substances is demonstrated at a solar panel plant where a common material silica sand is transformed into. In 1911, Rutherford said, the atom's majority part was possibly blank with the positive charges accumulated at the center. The atomic theory led to the creation of the law of multiple proportions. The contributions to atomic theory of Dalton, Proust, Lavoisier, as well as those of the Arabic scientist, Jbir ibn ayyn, who died in 803 AD, are discussed. As per the model, electrons are scattered around in the atom with a cloud of positive charge. Thompson modified the theory and proposed the plum pudding model. This lesson plan covers major developments and changes in atomic theory, with a focus on the 1800s and 1900s. In this study guide, you can revise how the periodic table arranges elements according to. He stated that atoms are further indivisible, and the atoms of an element are the same. Atoms are made from protons, neutrons and electrons. In 1803, John Dalton put forward the solid sphere model. The development of the atomic theory occurred in multiple stages. Around the year 400 B.C., a scientist named Democritus developed a theory stating that the smallest indivisible particle in the world existed. An individual can draw an Atomic Theory timeline diagram to understand the atomic theory modification. Many physicists put forward theories about the structure of atoms and molecules, contributing to the modifications of the Atomic theory. The word atom has come from the word atmos, which means uncut. This smallest particle of matter is called an atom. Scientists considered that when a matter gets broken continuously, it will end with the smallest particle, which can not be further divisible. Atoms are the particles present in the matter.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
